 Image 1 of 1
Image 1 of 1


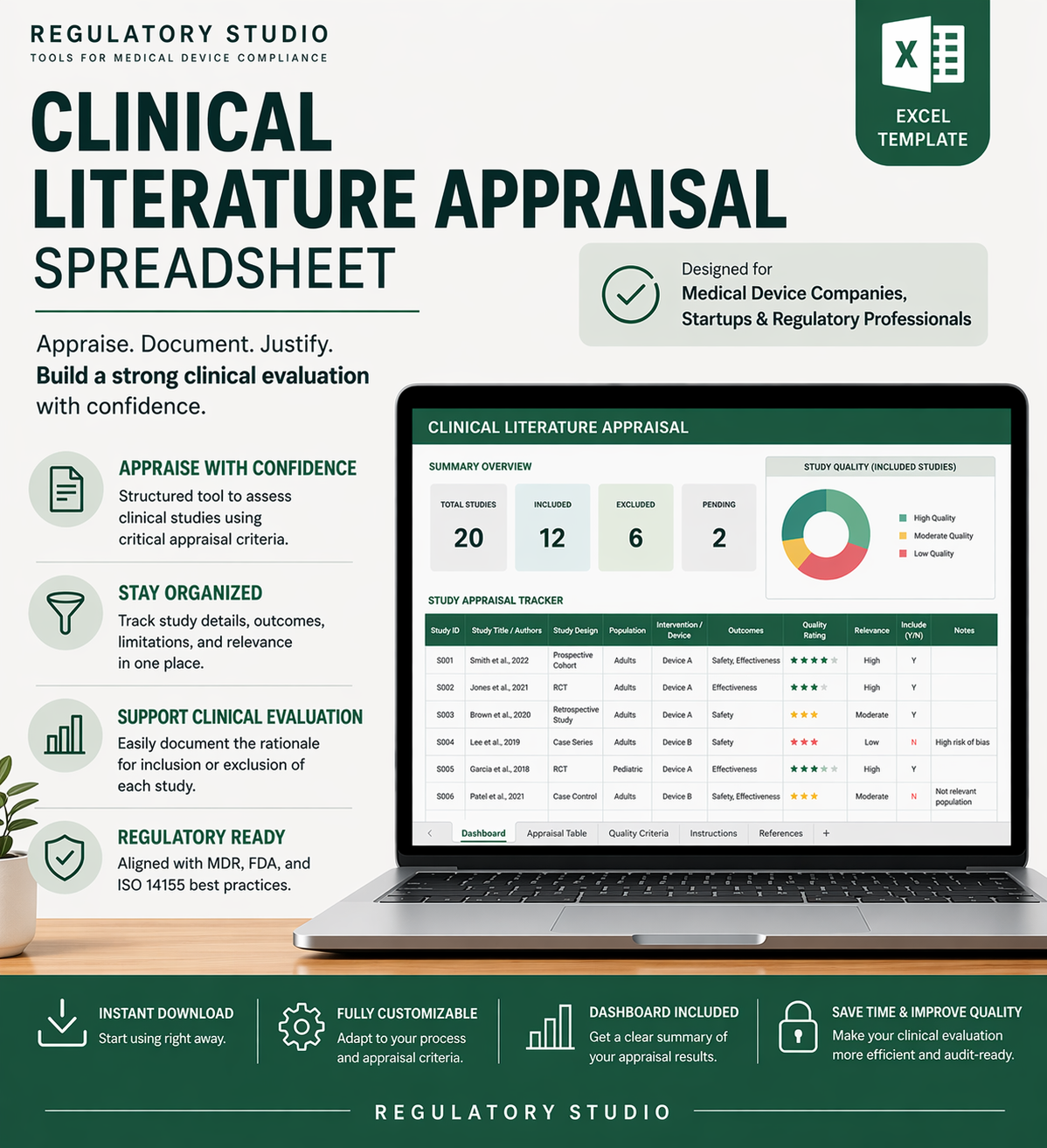
Clinical Literature Appraisal Spreadsheet
This spreadsheet is designed to organize and simplify your process of appraising clinical literature as part of your post-market surveillance activities or Clinical Evaluation Reports (CERs). It allows you to systematically capture key data points such as study design, patient population, outcomes, and relevance to your device. By structuring and standardizing this information, the spreadsheet helps ensure thorough and consistent evaluations, making it easier to identify trends, gaps, and risks.
This spreadsheet is designed to organize and simplify your process of appraising clinical literature as part of your post-market surveillance activities or Clinical Evaluation Reports (CERs). It allows you to systematically capture key data points such as study design, patient population, outcomes, and relevance to your device. By structuring and standardizing this information, the spreadsheet helps ensure thorough and consistent evaluations, making it easier to identify trends, gaps, and risks.



